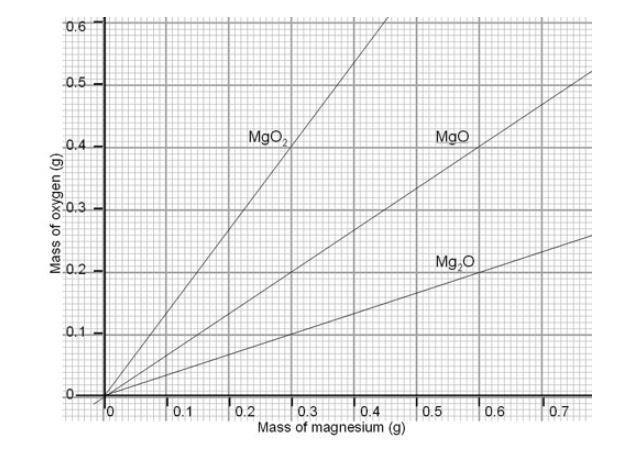
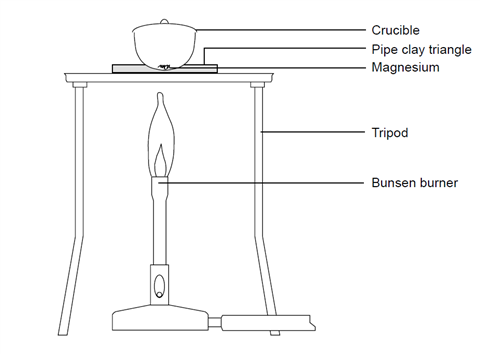
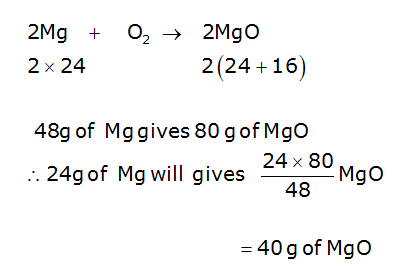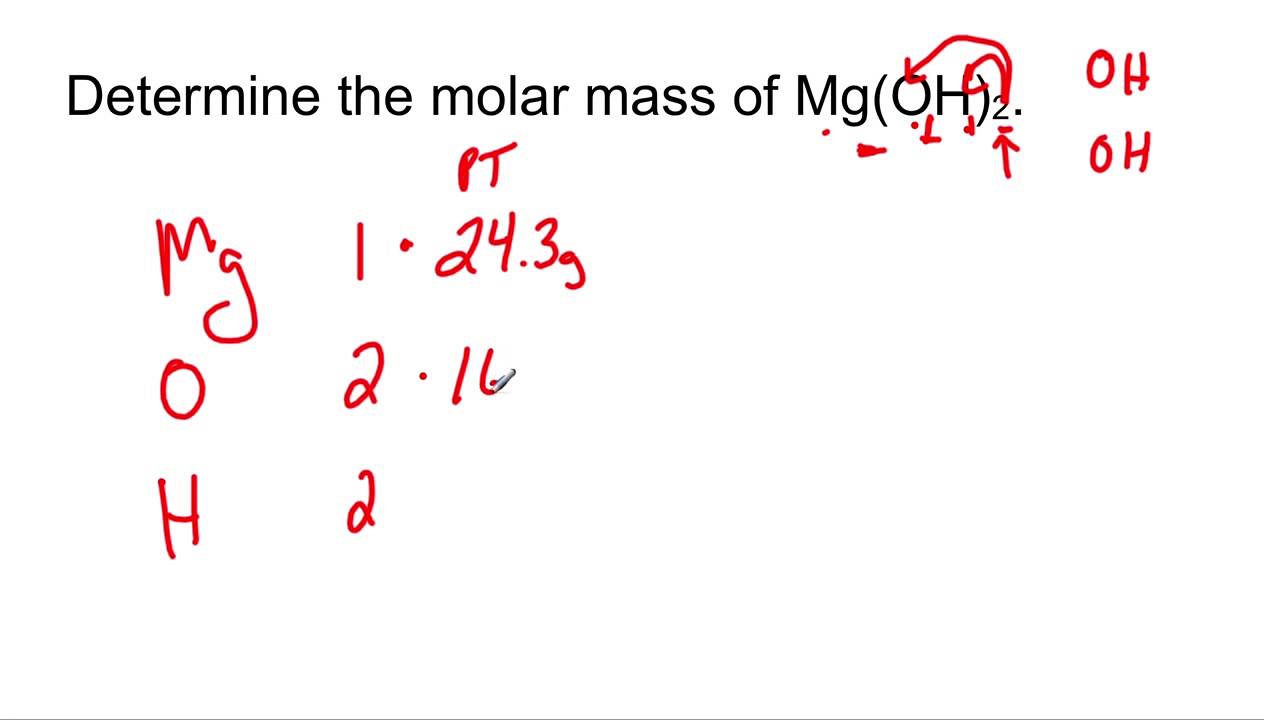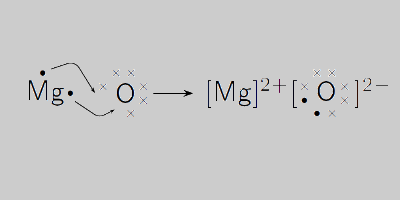A 3.250g sample of magnesium is burned in a container of 12.500g oxygen. What mass of oxygen gas remains unreacted after the magnesium has been completely consumed to form magnesium oxide as
![Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ] Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ]](https://dwes9vv9u0550.cloudfront.net/images/8090905/68318e51-f37d-486f-b3fb-f22fbc1043ce.jpg)
Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ]
![Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ] Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ]](https://dwes9vv9u0550.cloudfront.net/images/9335701/877be613-774e-48a4-af41-0d7e1c19f562.jpg)
Calculate the number of molecules present in 0.5 moles of magnesium oxide ( MgO) . [Atomic weights : Mg = 24, O = 16 ]

N. Calculate the number of moles ofmagnesium oxide, MgO in i. 80 g andii. 10 g of the compound. - Brainly.in

DOC) IB Chemistry IA: Determining the Empirical Formula of Magnesium Oxide | Josephine Yeh - Academia.edu

1a.calculate the relative formula mass of magnesium oxide when the relative atomic masses are O=16 - Brainly.com