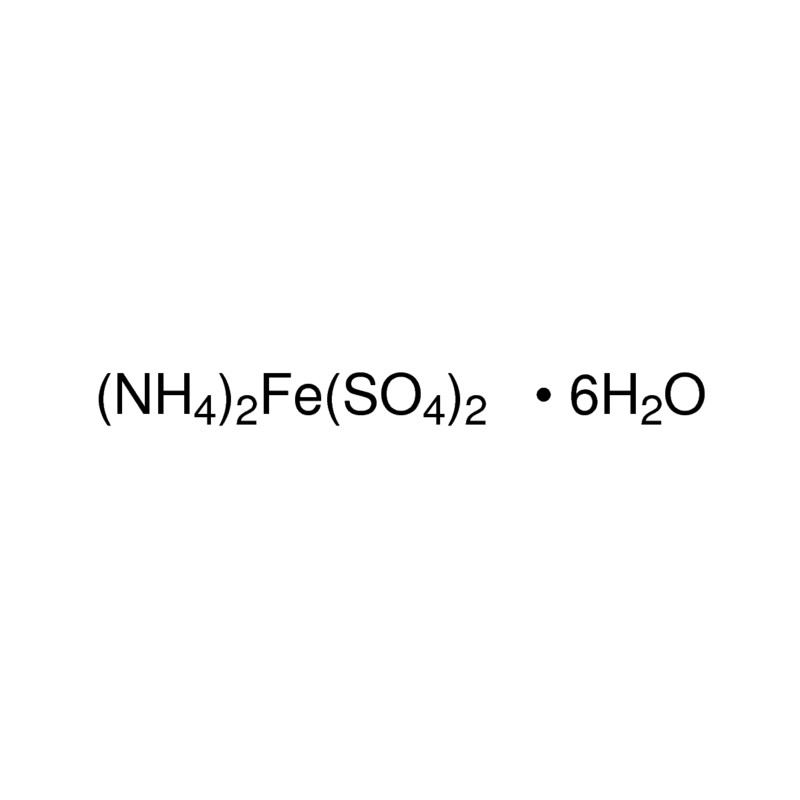Oxidation state of fe in mohr's salt with solution and formula Step wise explanation needed - Chemistry - Redox Reactions - 16519045 | Meritnation.com

The molecular formula of Mohr's salt is (NH.),SO.FeS0.6H,O(1) Find the number of atoms of each element.(2) - Brainly.in

The percentage composition of Mohr's salt is 14 32% Fe2+, 9 2% NH4+, 49% SO42- 27 57% H2O - Chemistry - Some Basic Concepts of Chemistry - 10320757 | Meritnation.com






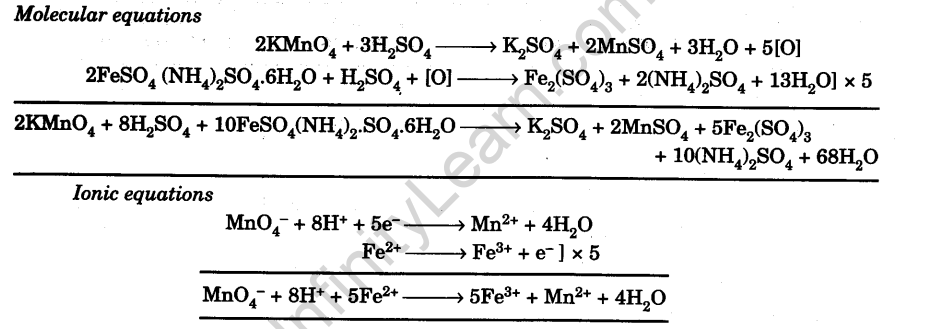



![Solved] Mohr's salt is Solved] Mohr's salt is](https://d10lpgp6xz60nq.cloudfront.net/ss/web/2041461.jpg)