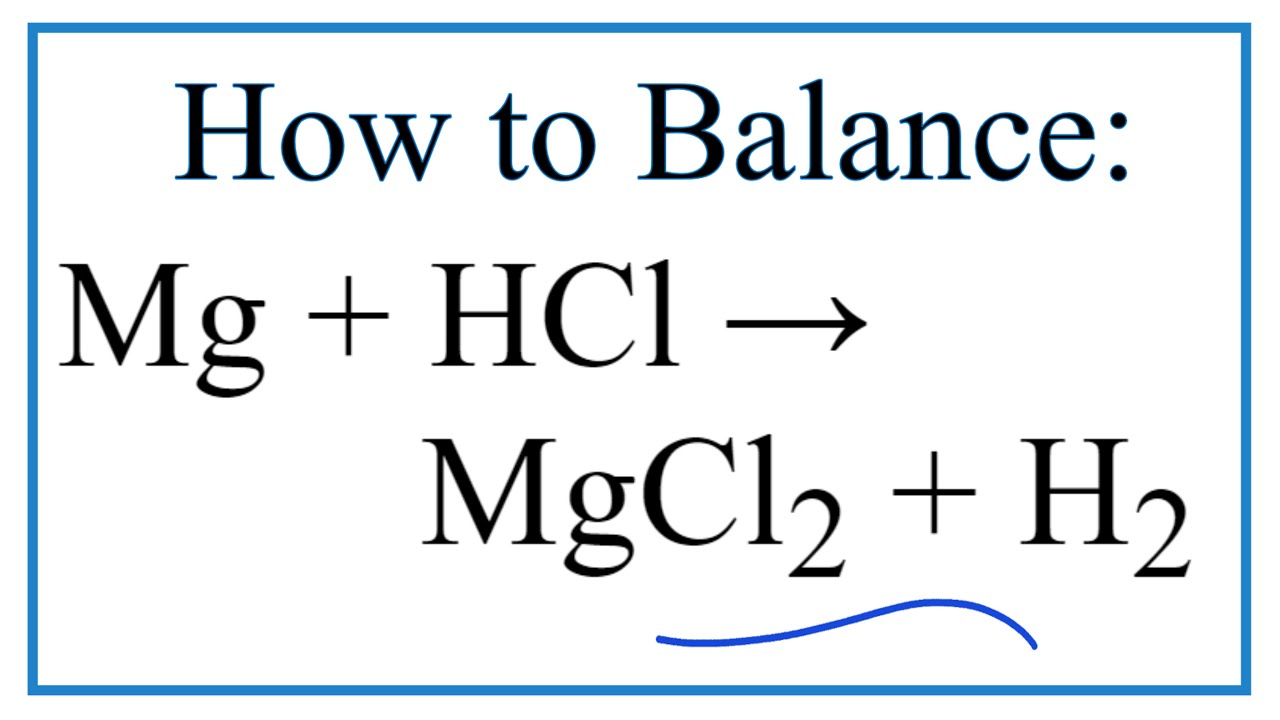
One gram of an alloy of aluminium and magnesium when heated with excess of dilute HCl forms magnesium chloride, aluminium chloride and hydrogen. The evolved hydrogen collected over mercury at 0^oC has

Question Video: Identifying the Name of the Gas Produced When Magnesium Metal Reacts with Hydrochloric Acid | Nagwa
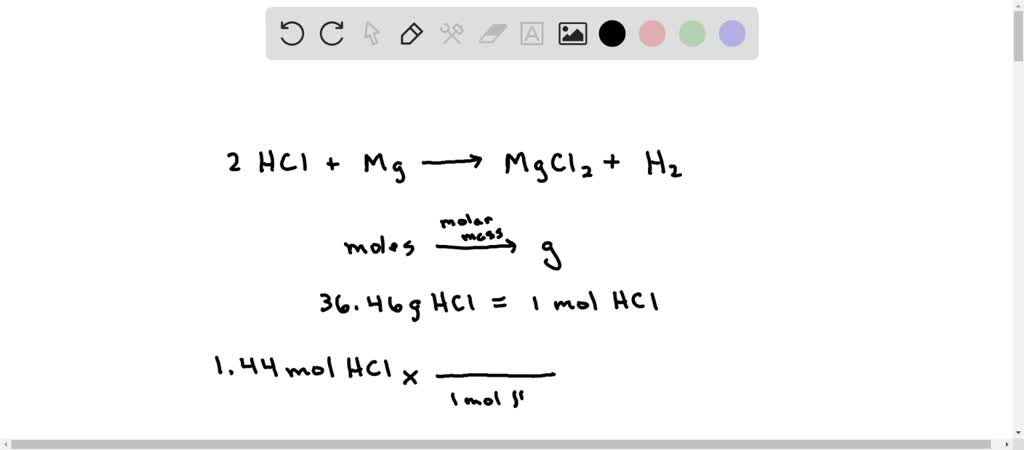
SOLVED: When magnesium metal (Mg) reacts with hydrochloric acid (HCl), magnesium chloride (MgCl2) and hydrogen gas (H2) are produced according to the following chemical equation. 2 HCl (aq) + Mg (s) →