acid base - What is the relative rate of diffusion of ammonia to hydrogen chloride, both in gaseous states? - Chemistry Stack Exchange
![Development of the diffusion rate of [BMIM]Cl at different coagulation... | Download Scientific Diagram Development of the diffusion rate of [BMIM]Cl at different coagulation... | Download Scientific Diagram](https://www.researchgate.net/profile/Yumei-Zhang-8/publication/251387805/figure/fig3/AS:669046437584897@1536524509156/Development-of-the-diffusion-rate-of-BMIMCl-at-different-coagulation-temperature-with.png)
Development of the diffusion rate of [BMIM]Cl at different coagulation... | Download Scientific Diagram

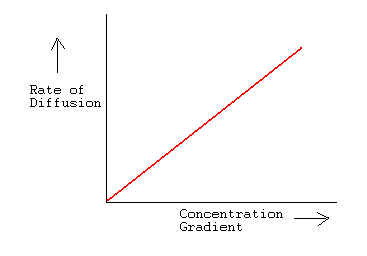
SOLVED:The rate of diffusion of a gas is proportional to: (a) (P)/(√(d)) (b) (P)/(d) (c) √((P)/(d)) (d) (√(P))/(d)

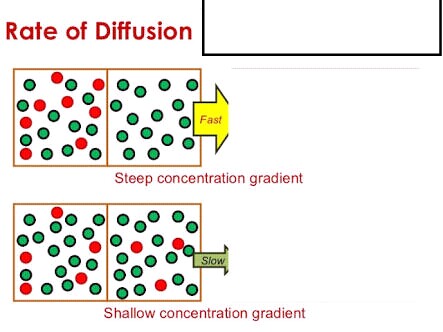
These substances are required to prove that the speed of diffusion is different for different substances.
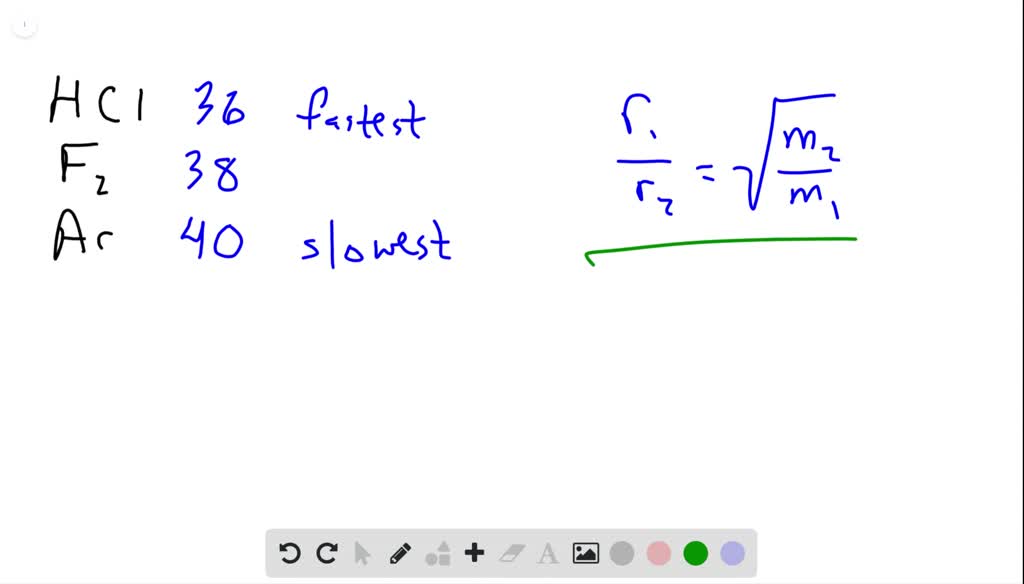
SOLVED:Rank the following gases in order of their speed of diffusion through a membrane, and calculate the ratio of their diffusion rates: HCl, F2, Ar .