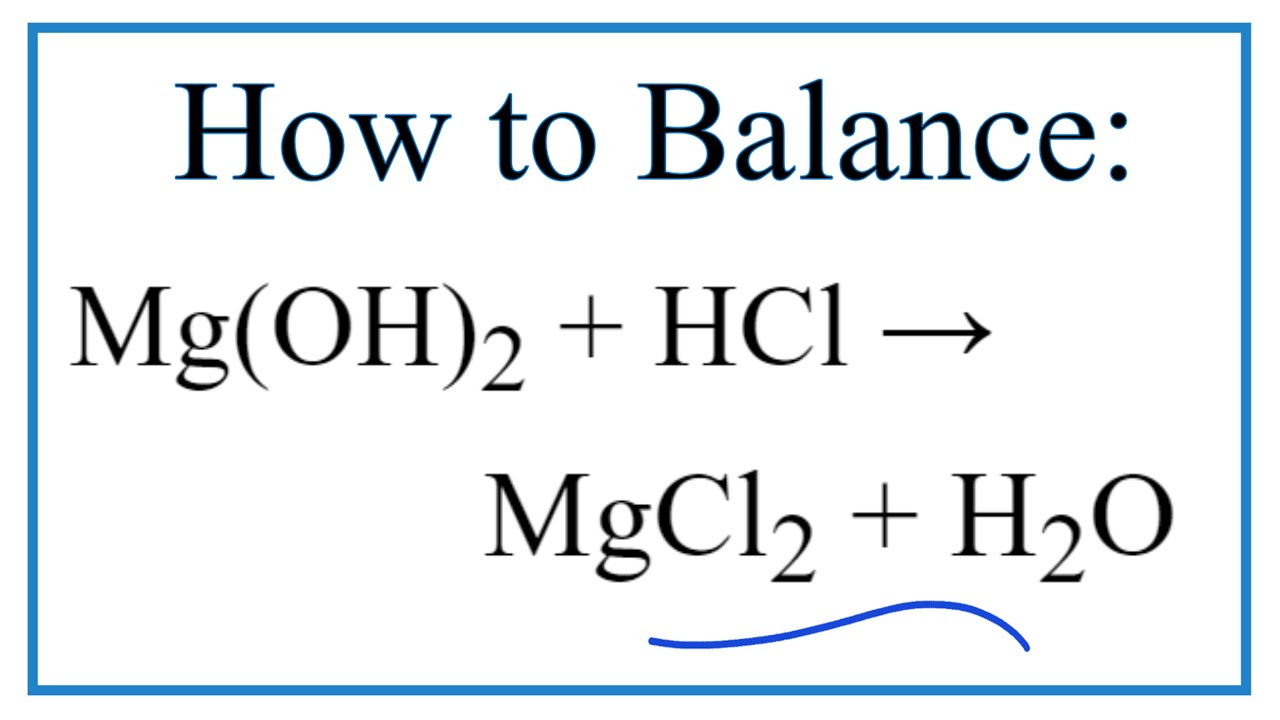
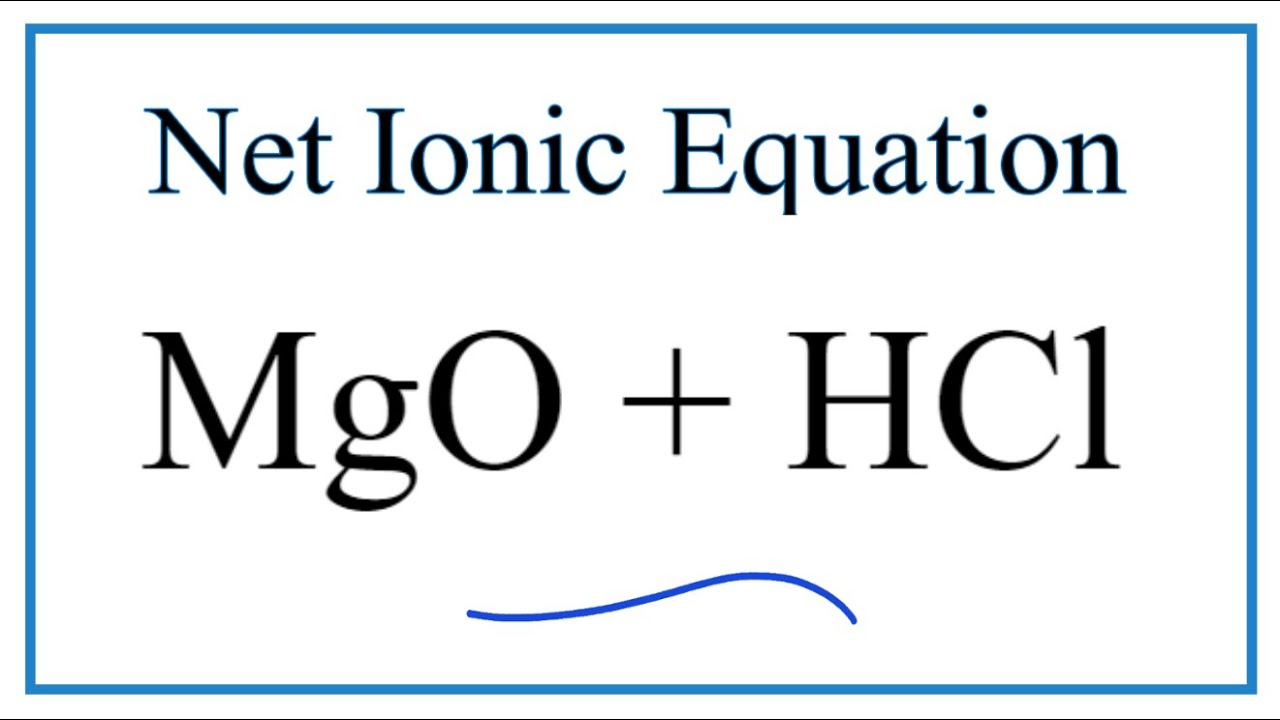Malox, an over-the-counter antacid, contains aluminum hydroxide, Al(OH) 3, and magnesium hydroxides, Mg(OH). What are balanced equations for the reaction of both with stomach acid (HCl)? - Quora
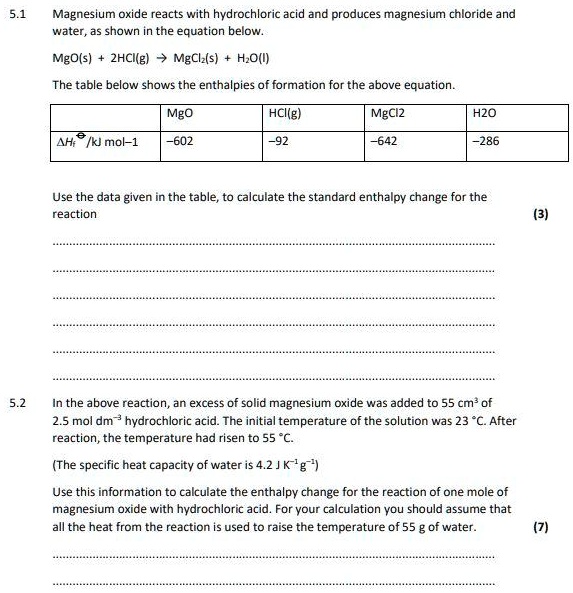
SOLVED: Magnesium oxide reacts with hydrochloric acid and produces magnesium chloride and water, as shown in the equation below: MgO(s) zhCIlg) MgCl (s) H,O(l) The table below shows the enthalpies of formation

SOLVED: Consider the reaction of magnesium oxide with hydrochloric acid: Mgo (s) 2 HCI (aq) + MgClz (aq) Hz (g) AH -151kI Is the reaction of MgO with HCl exothermic or endothermic?

16/10/2015 Acids and Bases Hydrochloric acidCitric acidWater An acid is a “proton donor”: A base is a “proton acceptor”: H Cl H +- H O Na H O - + Sodium. - ppt download

Introduction to Acids and Bases. Acid A substance that produces hydrogen ions, H + (aq), when it dissolves in water. Sour-tasting and good conductors. - ppt download

Ionic equations A chemical equation shows the number of atoms and molecules of the reactants and products. Also shows physical state of reactants and products. - ppt download

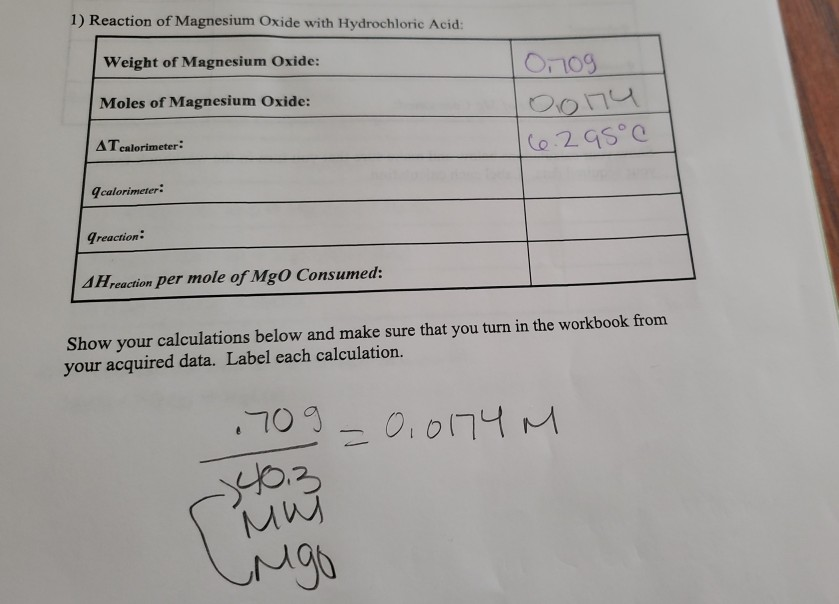
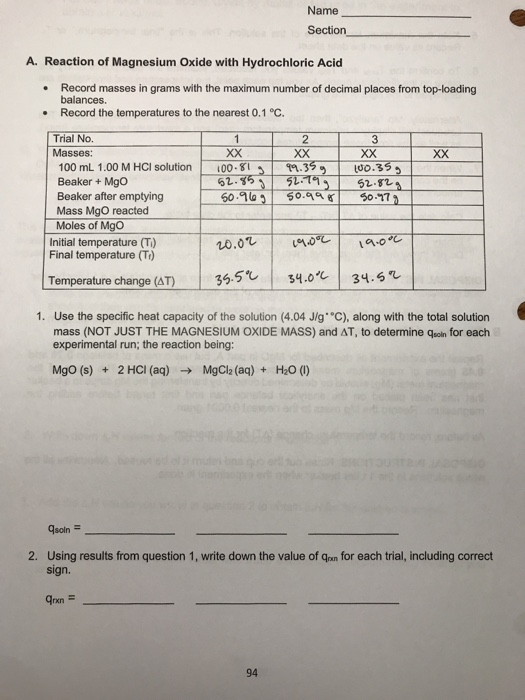
SOLVED: Reaction of Magnesium Oxide with Hydrochloric Acid Trial No Masses: XX XX 100 mL 00 M HCI solution Jno Ghs 91. 5S4 1o.31 Beaker Mgo 52 629 52 ,219 S2,6L) Beaker