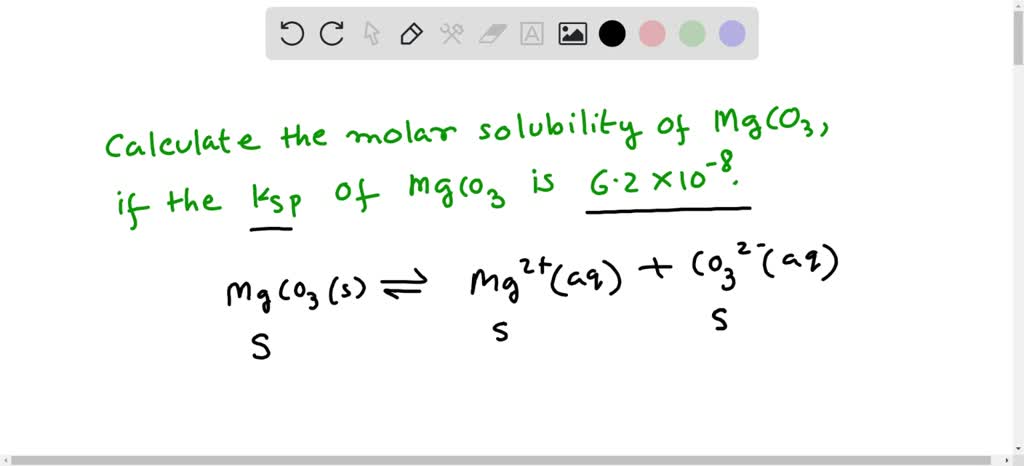Can you explain this sentence of my textbook: “It is because of high solubility product of mg(oh) 2 as compared to that of mg co3, that mg(OH) 2 is precipitated”? - Quora

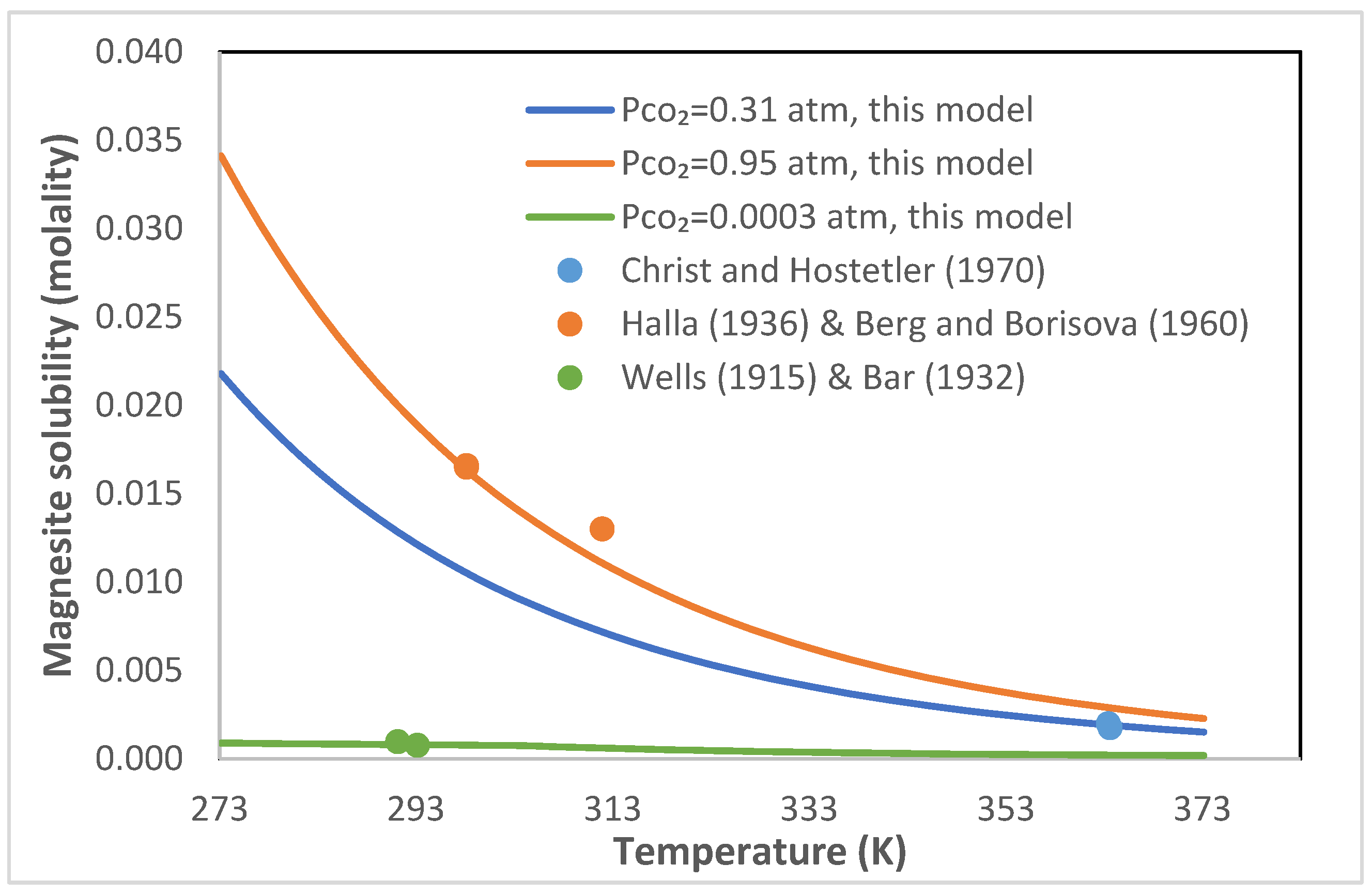
Solubilities of pure magnesium carbonates and hydroxide as a function... | Download Scientific Diagram

Table 1 from Mesoporous magnesium carbonate as a drug delivery vehicle for stabilising amorphous drugs and regulating their release rate | Semantic Scholar

SOLVED:Calculate The Ks p of magnesium carbonate (Mg CO3) is 2.6 ×10^-9 . What is the solubility of Mg CO3 in pure water?
The Kinetics of Magnesium Carbonate Crystallization for Traditional Salt Production Wastewater Recovery

Figure SI-1.3 Solubility of the magnesian calcites as a function of the... | Download Scientific Diagram
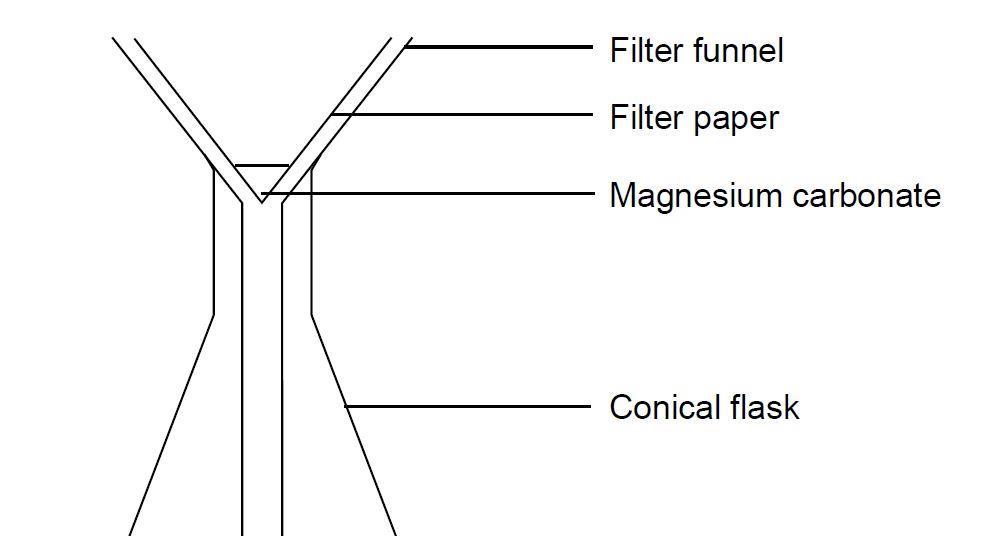
Making magnesium carbonate: the formation of an insoluble salt in water | Experiment | RSC Education

Table 14 from IUPAC-NIST Solubility Data Series. 95. Alkaline Earth Carbonates in Aqueous Systems. Part 1. Introduction, Be and Mg | Semantic Scholar
Phase stability and kinetics of methane hydrate formation in presence of calcium and magnesium carbonate

The molar solubility of magnesium carbonate is 1.87x10^-4 mol/L. Calculate the Ksp of MgCO3. - YouTube

SOLVED: 1) Use the solubility product constant Kap of magnesium carbonate to cakculate AG" for the reaction MgCOz(s) Mg2t(aq) + CO; ?(a9); Ky 6.8x10 ' Is this reaction reaclant- 6r product-favored? Why?
✓ Solved: Ksp for magnesium carbonate, MgCO3, has a value 3.5 × 10^-8 at 25^∘ C . Calculate the solubility...