Why is aniline treated with the excess of concentrated sulfuric acid in the preparation of sulfanilic acid? - Quora

Sulfuric Acid LO: Outline uses and reactions involving Sulfuric Acid Starter: What is an acid? - ppt download

Assertion: H2SO4 forms only one series of salts.Reasons: The molecules of H2SO4 consists of only one OH group.
Sodium carbonate reacts with dil. H2SO4 to give the respective salt, water and carbon dioxide. - Sarthaks eConnect | Largest Online Education Community

Question Video: Identifying the Salt That Produces Purple Fumes upon Addition of Sulfuric Acid | Nagwa

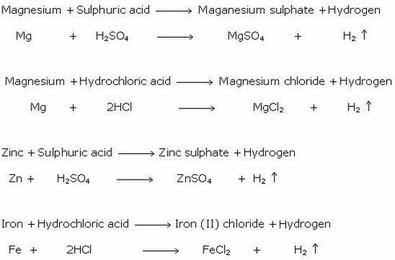
SOLVED: student wants to prepare a salt starting with H2SO4' Select all of the compound types that can react with HzSO4 to form salt salt acid acid salt basic oxide base metal
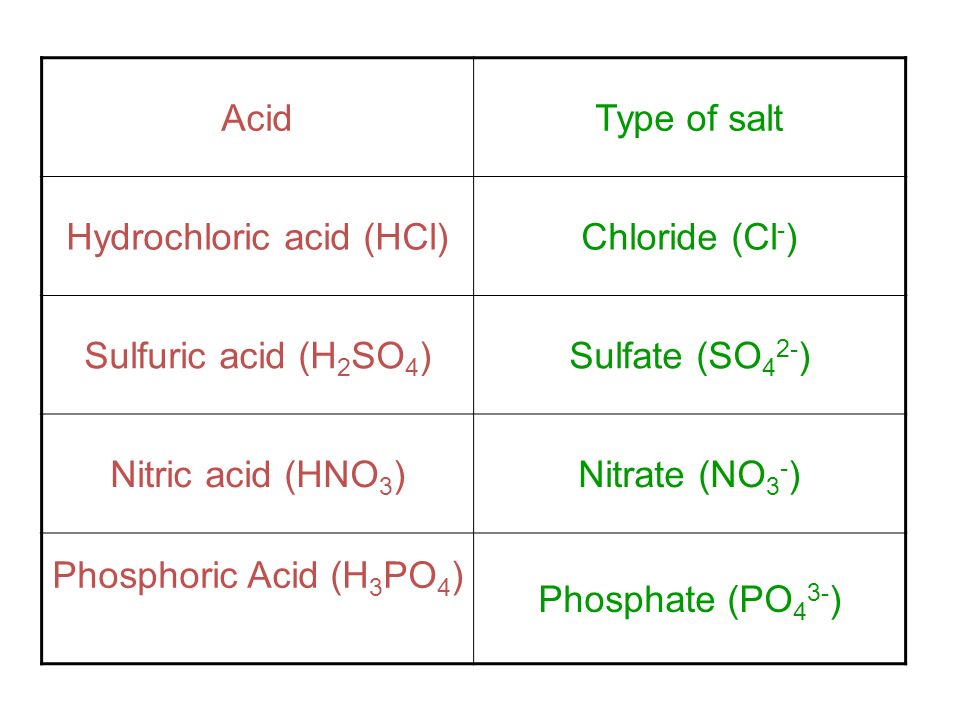
Sulphuric acid forms two types of salts with an alkali.Give reasons. - Sarthaks eConnect | Largest Online Education Community

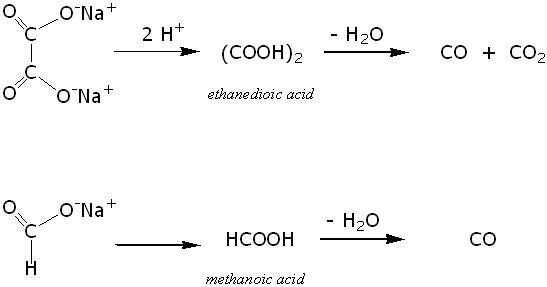
Sodium salt of an organic acid 'X' produces effervescence with conc.H2SO4 . 'X' reacts with the acidified aqueous CaCl2 solution to give a white precipitate which decolourizes acidic solution of KMnO4 'X'