CHARACTERISTICS OF METALS & NON-METALS ACCEPT THE CHALLENGE, SO YOU MAY FEEL THE EXHILARATION OF VICTORY. GEORGE S. PATTON Ch 17. J.C. Rowe Windsor University. - ppt download
What happens when calcium and magnesium react with water? What are the equations to explain it? - Quora

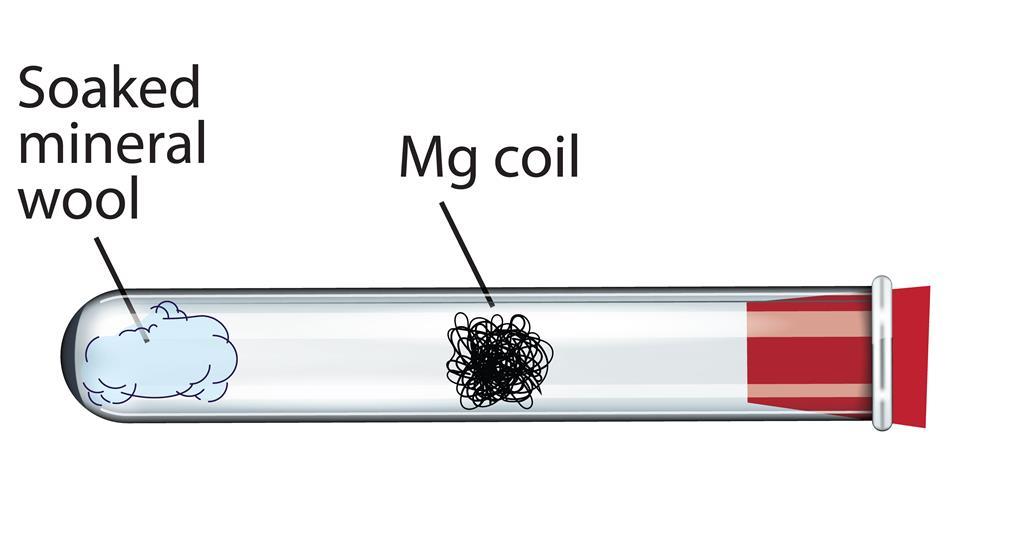
Metals react differently with water. The more vigorous is the reaction, the more is the bubble formation. The following diagram illustrates the reaction of sodium, magnesium and copper with water.What can be